SOLDERING PRODUCTS
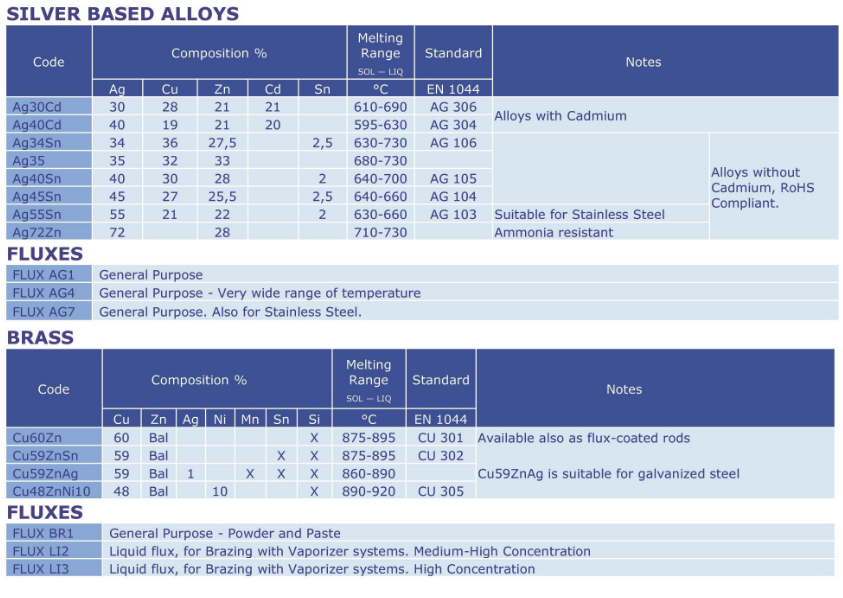
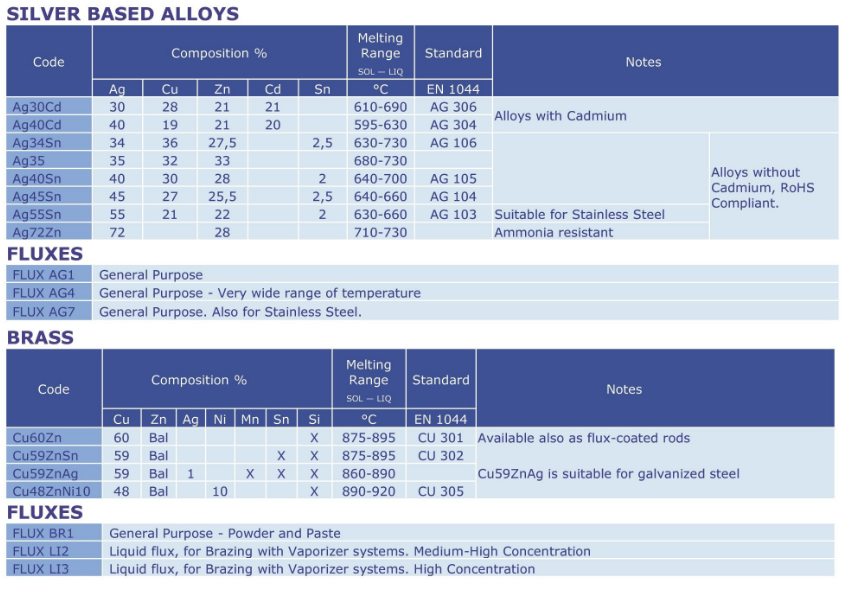
Silver based alloys Cadmium free
General purpose brazing alloys based on Silver.Products can be divided into two sections, those with Tin and those without Tin.
These alloys do not contain Cadmium. They overcome the Health and Safety problems
associated with “Cadmium alloys” and are therefore compliant with RoHS regulation.
Products are supplied as bare rods, or as coated rods.
Silver based alloys Cadmium free
General purpose brazing alloys based on Silver.Products can be divided into two sections, those with Tin and those without Tin.
These alloys do not contain Cadmium. They overcome the Health and Safety problems
associated with “Cadmium alloys” and are therefore compliant with RoHS regulation.
Products are supplied as bare rods, or as coated rods.
Silver based alloys with Cadmium
General purpose brazing alloys based on Silver and containing Cadmium.
Cadmium and its oxides are toxic, therefore special safety precautions must be followed during brazing operations (e.g. mechanical ventilation and/or respiratory masks). The use of these alloys in applications
where food, beverages and/or human health in general is involved is forbidden.
Products are supplied as bare rods, or as coated rods.
General purpose brazing alloys based on Silver and containing Cadmium.
Cadmium and its oxides are toxic, therefore special safety precautions must be followed during brazing operations (e.g. mechanical ventilation and/or respiratory masks). The use of these alloys in applications
where food, beverages and/or human health in general is involved is forbidden.
Products are supplied as bare rods, or as coated rods.
Silver based alloys Special applications
Thanks to the addition of specific elements or to their particular composition these Silver alloys have improved characteristics that make them suitable for special applications or to operate in particular
condition.
Thanks to the addition of specific elements or to their particular composition these Silver alloys have improved characteristics that make them suitable for special applications or to operate in particular
condition.
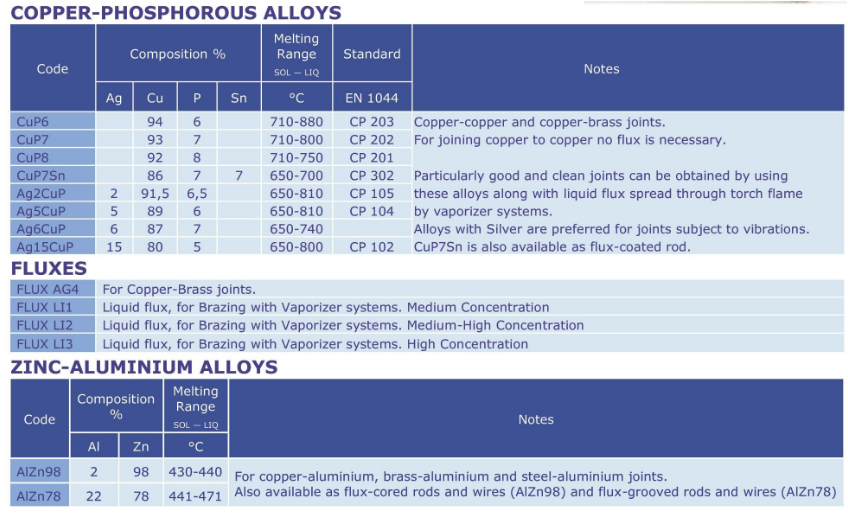
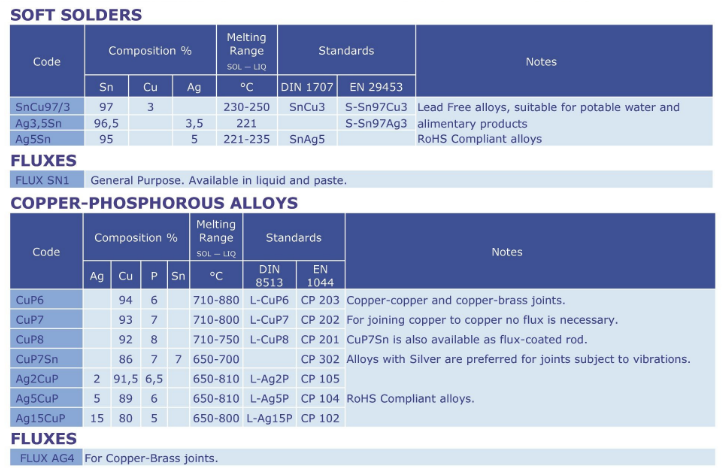
Copper Phosphorous alloys
Brazing alloys with Phosphorous.
These low-cost alloys have self-fluxing properties (when used on copper).
They are divided into groups containing Silver and a group without Silver.
Copper-Phosphorous alloys are used to join copper and copper-alloy base
metals (brass, bronzes). They are not used to join ferrous or nickel-based
base metals.
Only CuP7Sn are available in a coated rod, the rest is only available as bare rods.
Brazing alloys with Phosphorous.
These low-cost alloys have self-fluxing properties (when used on copper).
They are divided into groups containing Silver and a group without Silver.
Copper-Phosphorous alloys are used to join copper and copper-alloy base
metals (brass, bronzes). They are not used to join ferrous or nickel-based
base metals.
Only CuP7Sn are available in a coated rod, the rest is only available as bare rods.
Copper based and Bronze alloys for Brazing
There are four categories of brazing alloys:
General Purpose Brasses
Brasses with addition of Silver and/or Nickel, with improved characteristics
High Temperature and Special Applications Brasses
Pure Copper for furnace brazing
These alloys are economical to use and cover from large joint gaps to free flowing pure copper.
There are four categories of brazing alloys:
General Purpose Brasses
Brasses with addition of Silver and/or Nickel, with improved characteristics
High Temperature and Special Applications Brasses
Pure Copper for furnace brazing
These alloys are economical to use and cover from large joint gaps to free flowing pure copper.
Aluminium and Magnesium based alloys
Aluminium based alloys are for TIG / MIG techniques. They are used to join
aluminium base metals; some alloys are also suitable for the Brazing technique.
Aluminium based alloys are for TIG / MIG techniques. They are used to join
aluminium base metals; some alloys are also suitable for the Brazing technique.
Soldering Process
Soldering is a process by which two metals or alloys are joined together with
a third metal or alloy. The third metal or alloy has a much lower melting point
compared to the first two metals. Soldering is different from adhesive joining.
Adhesives bond by mechanical attraction. In soldering, there is also a chemical
reaction in addition to physical reaction.
Soldering is primarily used to provide a metallurgical joint with good
electrical contact and/or seal against leakage. Solders typically do not provide
high mechanical strength, given the soft nature of popular solder materials.
Soldering is used extensively in the electronics industry and printed circuit
boards. It is also used in joining metals in industries such as cutlery, tools,
metal box making etc.
In order to have a good soldering joint, one must form inter-metallic layers
between the solder material and the base metal. Solder that simply solidifies
over the base metal without forming any bonds are very weak in mechanical
strength and also poorly conductive.
Within each inter-metallic layer, there are actually a number of different
compounds formed by the solder materials and the base metal. These compounds are typically quite brittle and will adversely affect the integrity of the solder joint. As the joint is subject to stress, thermal cycles, vibration, or shock, the inter-metallic layers are usually where it starts to fail. Since the inter-metallic layers are inevitable, it is best to keep it as thin as possible.
We have to solder fast. This requires knowing the properties of the base
metal and the speed it dissolves in the solder material.
Tin based solders, in particular, are especially aggressive dissolving metals. The
figure below illustrates the approximate dissolution rates of a few typical base
metals in tin
From the above it is clear that Ni has one of the lowest dissolution rates.
So it is not surprising that nickel is frequently used as a barrier for soldering.
Inter-metallic layer is a essential to create a strong soldering joint.
Once created, it grows at any temperature and accelerates exponentially as the
temperature goes higher. This growth continues until the entire joint is
occupied by the inter-metallic compounds, or either the base metal or the
solder is exhausted. Thus the rules of soldering are:
Remember, the inter-metallic layer grows at any temperature, the higher
the faster.
Applications
Soldering is a process by which two metals or alloys are joined together with
a third metal or alloy. The third metal or alloy has a much lower melting point
compared to the first two metals. Soldering is different from adhesive joining.
Adhesives bond by mechanical attraction. In soldering, there is also a chemical
reaction in addition to physical reaction.
Soldering is primarily used to provide a metallurgical joint with good
electrical contact and/or seal against leakage. Solders typically do not provide
high mechanical strength, given the soft nature of popular solder materials.
Soldering is used extensively in the electronics industry and printed circuit
boards. It is also used in joining metals in industries such as cutlery, tools,
metal box making etc.
In order to have a good soldering joint, one must form inter-metallic layers
between the solder material and the base metal. Solder that simply solidifies
over the base metal without forming any bonds are very weak in mechanical
strength and also poorly conductive.
Within each inter-metallic layer, there are actually a number of different
compounds formed by the solder materials and the base metal. These compounds are typically quite brittle and will adversely affect the integrity of the solder joint. As the joint is subject to stress, thermal cycles, vibration, or shock, the inter-metallic layers are usually where it starts to fail. Since the inter-metallic layers are inevitable, it is best to keep it as thin as possible.
We have to solder fast. This requires knowing the properties of the base
metal and the speed it dissolves in the solder material.
Tin based solders, in particular, are especially aggressive dissolving metals. The
figure below illustrates the approximate dissolution rates of a few typical base
metals in tin
From the above it is clear that Ni has one of the lowest dissolution rates.
So it is not surprising that nickel is frequently used as a barrier for soldering.
Inter-metallic layer is a essential to create a strong soldering joint.
Once created, it grows at any temperature and accelerates exponentially as the
temperature goes higher. This growth continues until the entire joint is
occupied by the inter-metallic compounds, or either the base metal or the
solder is exhausted. Thus the rules of soldering are:
- Solder as quickly as possible.
- Use the lowest possible soldering temperature
that yields acceptable joints. - Avoid repeated soldering just to make the joint look better.
Added exposure to high temperatures only increases the inter-metallic
layer. - The joint may look pretty but is indeed weaker.
- Avoid elevated operating temperatures for the end product. Keep it well below
the soldering temperature as much as possible.
Remember, the inter-metallic layer grows at any temperature, the higher
the faster.
Applications
- Heat Exchangers
- Industrial and home refrigerators
- Heating, Cooling and Air
Conditioning – Installation - Radiators
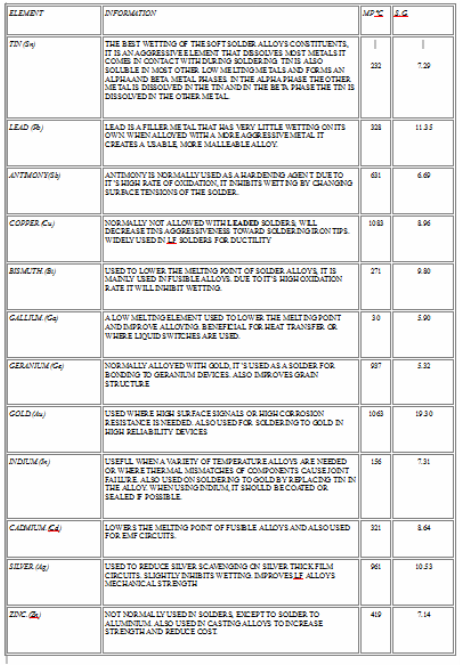
Metals in Solders Alloys
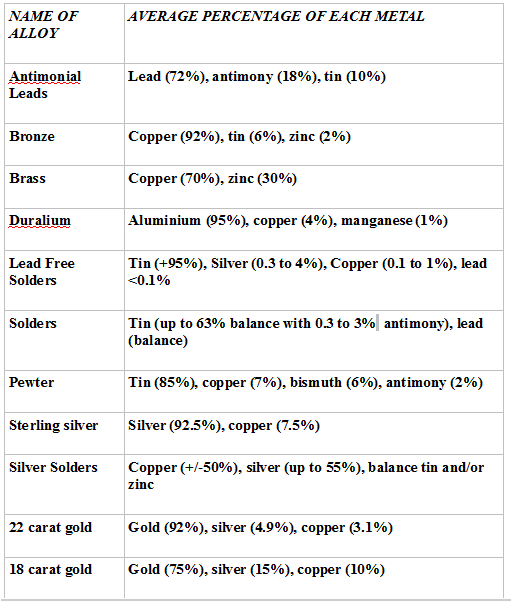
Non Ferrous Alloy Types