Welding & Brazing & Soldering
Basic difference between the three processes lies in the twin factor of requirement specifications of the desired result and the chemical and physical properties of the base metals at hand. Brazing is a robust process, conducted at a higher temperature compared to soldering, and gives far stronger joints as compared to any other metal bonding process, leave alone soldering. Soldering is done at lower temperatures and therefore can't produce strong joints. In situations where strength is not the key joint characteristic that manufacturer is looking at, soldering is more efficient a process.
Another difference is in what kind of base metal is to be joined. The physical dimensions of the base metals should be paid attention to while deciding on the suitable bonding method. Apart from the above mentioned differences, there are other differences also which are numerated below:
Basic difference between the three processes lies in the twin factor of requirement specifications of the desired result and the chemical and physical properties of the base metals at hand. Brazing is a robust process, conducted at a higher temperature compared to soldering, and gives far stronger joints as compared to any other metal bonding process, leave alone soldering. Soldering is done at lower temperatures and therefore can't produce strong joints. In situations where strength is not the key joint characteristic that manufacturer is looking at, soldering is more efficient a process.
Another difference is in what kind of base metal is to be joined. The physical dimensions of the base metals should be paid attention to while deciding on the suitable bonding method. Apart from the above mentioned differences, there are other differences also which are numerated below:
- Strength wise, brazing gives far stronger results than soldering.
- Procedural flexibility is comparatively higher in brazing than in soldering.
- Comparing soldering and brazing on cost efficacy criteria is quite difficult, as both of them are almost the same
- Soldering allows a certain degree of control over the process, but brazing allows a far better control over the process
Brazing
Brazing is a metal-joining process whereby a filler metal is heated above and distributed between two or more close-fitting parts by capillary action. The filler metal is brought slightly above its melting (liquids) temperature while protected by a suitable atmosphere, usually a flux. It then flows over the base metal (known as wetting) and is then cooled to join the work pieces together.
Silver Soldering
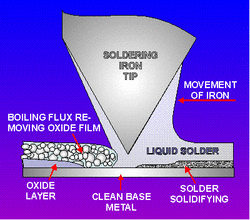
· Soldering is a process in which two or more metal items are joined together by melting and flowing a filler metal into the joint, the filler metal having a relatively low melting point. Soft soldering is characterized by the melting point of the filler metal, which is below 400 °C (752 °F).The filler metal used in the process is called solder.
· Soldering is distinguished from brazing by use of a lower melting-temperature filler metal; it is distinguished from welding by the base metals not being melted during the joining process. In a soldering process, heat is applied to the parts to be joined, causing the solder to melt and be drawn into the joint by capillary and to bond to the materials to be joined by wetting. After the metal cools, the resulting joints are not as strong as the base metal, but have adequate strength, electrical conductivity, and water-tightness for many uses. Soldering is an ancient technique mentioned in the Bible and there is evidence that it was employed up to 5000 years ago in Mesopotamia.
Brazing is a metal-joining process whereby a filler metal is heated above and distributed between two or more close-fitting parts by capillary action. The filler metal is brought slightly above its melting (liquids) temperature while protected by a suitable atmosphere, usually a flux. It then flows over the base metal (known as wetting) and is then cooled to join the work pieces together.
Silver Soldering
· Soldering is a process in which two or more metal items are joined together by melting and flowing a filler metal into the joint, the filler metal having a relatively low melting point. Soft soldering is characterized by the melting point of the filler metal, which is below 400 °C (752 °F).The filler metal used in the process is called solder.
· Soldering is distinguished from brazing by use of a lower melting-temperature filler metal; it is distinguished from welding by the base metals not being melted during the joining process. In a soldering process, heat is applied to the parts to be joined, causing the solder to melt and be drawn into the joint by capillary and to bond to the materials to be joined by wetting. After the metal cools, the resulting joints are not as strong as the base metal, but have adequate strength, electrical conductivity, and water-tightness for many uses. Soldering is an ancient technique mentioned in the Bible and there is evidence that it was employed up to 5000 years ago in Mesopotamia.
Brazing vs. Welding
When you want to join metal parts, you can choose from several options–including adhesive bonding and mechanical fasteners–but when you want to create robust, permanent metal joints, you typically have only two choices: welding or brazing. While welding creates metal joints by applying concentrated heat at the joint to melt and fuse metals together, brazing involves significantly lower temperatures and does not entail the melting of base metals. Instead, a filler metal is melted and forced to flow into the joint through capillary action. (Welding usually adds a filler material as well.)
In welding and brazing, the strength of the joint often surpasses that of the base materials. But because the heat used in brazing is less intense, this process does not alter most physical properties and minimizes distortion, warping and stresses in the joint area. Additionally, brazing’s lower temperatures translate to less energy requirements.
But of course, the decision to weld or to braze comes down to the specifics of the application. Here are the factors to consider:
Size of the assembly: If you want to join large assemblies, welding is the more appropriate method. The tendency of larger assemblies to disperse heat can be a stumbling block for brazing, making it hard to reach the temperatures required for the filler metal to flow. In contrast, the concentrated heat of welding and its ability to trace a joint allow it to excel in joining big assemblies.
Thickness: Both methods are sound choices when metal sections are on the thick side, around 0.5 inches or more. Brazing has the edge, however, when it comes to thin sections. The high heat of welding can warp or burn through such sections. In contrast, brazing can help you avoid distortion.
Shape of joint: While both welding and brazing can create spot joints, the localized heat of welding offers the advantage of speed and low cost. However, when it comes to linear joints, the manual tracing required by welding makes it the less convenient choice. Additionally, brazing can just as easily draw the filler metal into straight, curved or irregular joint configurations.
Types of materials: Brazing soundly beats welding when joining dissimilar metals. As long as the filler material is metallurgically compatible with both base metals and melts at a lower temperature, brazing can create strong joints with barely any alteration of the base metals’ properties. In contrast, base materials are melted during welding so joining two dissimilar metals using this method can entail complex and costly techniques.
Production volume: In manual jobs, the above factors–size, thickness, shape of joint and materials–will guide your decisions, but when part volumes are in the hundreds or thousands, production techniques and cost will become the most important considerations. While both brazing and welding can be automated, brazing allows for more degrees of automation. With welding, you’re usually forced to choose between two extremes–either weld manually, one by one, or use pricey, cutting-edge equipment for large runs. In contrast, brazing can support medium-sized runs.
Appearance: With a neat and discreet strip, brazed joints are generally more attractive than welded ones, which have non-uniform beads. The vast majority of the time, you won’t need additional finishing operations after brazing.
Welding joins metals by melting and fusing them together, usually with the addition of a welding filler metal. The joints produced are strong, usually as strong as the metals joined or even stronger. In order to fuse the metals, a concentrated heat is applied directly to the joint area. This heat is high temperature. It must be - in order to melt the "base" metals (the metals being joined) and the filler metals as well.
When you want to join metal parts, you can choose from several options–including adhesive bonding and mechanical fasteners–but when you want to create robust, permanent metal joints, you typically have only two choices: welding or brazing. While welding creates metal joints by applying concentrated heat at the joint to melt and fuse metals together, brazing involves significantly lower temperatures and does not entail the melting of base metals. Instead, a filler metal is melted and forced to flow into the joint through capillary action. (Welding usually adds a filler material as well.)
In welding and brazing, the strength of the joint often surpasses that of the base materials. But because the heat used in brazing is less intense, this process does not alter most physical properties and minimizes distortion, warping and stresses in the joint area. Additionally, brazing’s lower temperatures translate to less energy requirements.
But of course, the decision to weld or to braze comes down to the specifics of the application. Here are the factors to consider:
Size of the assembly: If you want to join large assemblies, welding is the more appropriate method. The tendency of larger assemblies to disperse heat can be a stumbling block for brazing, making it hard to reach the temperatures required for the filler metal to flow. In contrast, the concentrated heat of welding and its ability to trace a joint allow it to excel in joining big assemblies.
Thickness: Both methods are sound choices when metal sections are on the thick side, around 0.5 inches or more. Brazing has the edge, however, when it comes to thin sections. The high heat of welding can warp or burn through such sections. In contrast, brazing can help you avoid distortion.
Shape of joint: While both welding and brazing can create spot joints, the localized heat of welding offers the advantage of speed and low cost. However, when it comes to linear joints, the manual tracing required by welding makes it the less convenient choice. Additionally, brazing can just as easily draw the filler metal into straight, curved or irregular joint configurations.
Types of materials: Brazing soundly beats welding when joining dissimilar metals. As long as the filler material is metallurgically compatible with both base metals and melts at a lower temperature, brazing can create strong joints with barely any alteration of the base metals’ properties. In contrast, base materials are melted during welding so joining two dissimilar metals using this method can entail complex and costly techniques.
Production volume: In manual jobs, the above factors–size, thickness, shape of joint and materials–will guide your decisions, but when part volumes are in the hundreds or thousands, production techniques and cost will become the most important considerations. While both brazing and welding can be automated, brazing allows for more degrees of automation. With welding, you’re usually forced to choose between two extremes–either weld manually, one by one, or use pricey, cutting-edge equipment for large runs. In contrast, brazing can support medium-sized runs.
Appearance: With a neat and discreet strip, brazed joints are generally more attractive than welded ones, which have non-uniform beads. The vast majority of the time, you won’t need additional finishing operations after brazing.
Welding joins metals by melting and fusing them together, usually with the addition of a welding filler metal. The joints produced are strong, usually as strong as the metals joined or even stronger. In order to fuse the metals, a concentrated heat is applied directly to the joint area. This heat is high temperature. It must be - in order to melt the "base" metals (the metals being joined) and the filler metals as well.
So welding temperatures start at the melting point of the base metals. Because welding heat is intense, it is impractical to apply it uniformly over a broad area. Welding heat is typically localized, pinpointed heat. This has its advantages. For example, if you want to join two small strips of metal at a single point, an electrical resistance welding setup is very practical.
This is a fast, economical way to make strong, permanent joints by the hundreds and thousands. However, if the joint is linear, rather than pinpointed, problems arise. The localized heat of welding tends to become a disadvantage. For example, suppose you want to butt-weld two pieces of metal - start by beveling the edges of the metal pieces to allow room for the welding filler metal. Then weld, first heating one end of the joint area to melting temperature, then slowly traveling the heat along the joint line, depositing filler metal in synchronization with the heat. This is a typical conventional welding operation. Let's look at its characteristics.
It offers one big plus - strength. Properly made, the welded joint is at least as strong as the metals joined. But there are minuses to consider. The joints are made at high temperatures, high enough to melt both base metals and filler metal. High temperatures can cause problems, such as possible distortion and warping of the base metals or stresses around the weld area. These dangers are minimal when the metals being joined are thick. But they may become problems when the base metals are thin sections. High temperatures are expensive as well since heat is energy, and energy costs money. The more heat you need to make the joint, the more the joint will cost to produce. Now consider the automated process.
What happens when you join not one assembly, but hundreds or thousands of assemblies. Welding, by its nature, presents problems in automation. We know that a resistance weld joint made at a single point is relatively easy to automate. But once the point becomes a line - a linear joint - the line has to be traced. It's possible to automate this tracing operation, moving the joint line, for example, past a heating station and feeding filler wire automatically from big spools. But this is a complex and exacting setup, warranted only when you have large production runs of identical parts.
Of course, welding techniques continually improve. You can weld on a production basis by electron beam, capacitor discharge, friction and other methods. But these sophisticated processes usually call for specialized and expensive equipment and complex, time consuming setups. They're seldom practical for shorter production runs, changes in assembly configuration or - in short - typical day-to-day metal joining requirements.
Oxyacetylene can be used to braze and to braze weld (bronze weld). These techniques are often confused, as they involve the use of the same equipment.
Bronze filler rod is an alloy composed of copper and tin. Brass filler rod is also an alloy, composed of copper and zinc. Either rod can be used.
The steel tube and steel plate have been heated to a dull red heat. The ‘bronze’ filler rod (coated with flux) is then introduced to the joint. It flows onto the two surfaces, forming a ‘fillet’ around the joint, joining the two surfaces.
Braze / bronze welding provides a strong joint and is ideal of steel frames, where a certain amount of flexibility is required.
The filler rod is broader than that used in brazing.
When brazing, flux is applied to the joint, to prevent oxidation of the surfaces. The steel tube and plate are heated to bright red heat and the brazing rod (filler rod) applied to the joint. The end of the filler rod melts and flows around the joint, through capillary attraction.
This gives a joint that looks like a ‘line’ of bronze.
Brazing is ideal for manufacture of bicycle frames, as it does not ‘fracture’ as easily as steel welded joints.
Brazing and braze welding (bronze welding), can be used to join similar and also dissimilar metals such as:
Mild Steel - Galvanized Steel
Stainless Steel - Copper
Mild Steel - Stainless Steel
Stainless Steel - Copper-Nickel
Mild Steel - Cast Iron
This is a fast, economical way to make strong, permanent joints by the hundreds and thousands. However, if the joint is linear, rather than pinpointed, problems arise. The localized heat of welding tends to become a disadvantage. For example, suppose you want to butt-weld two pieces of metal - start by beveling the edges of the metal pieces to allow room for the welding filler metal. Then weld, first heating one end of the joint area to melting temperature, then slowly traveling the heat along the joint line, depositing filler metal in synchronization with the heat. This is a typical conventional welding operation. Let's look at its characteristics.
It offers one big plus - strength. Properly made, the welded joint is at least as strong as the metals joined. But there are minuses to consider. The joints are made at high temperatures, high enough to melt both base metals and filler metal. High temperatures can cause problems, such as possible distortion and warping of the base metals or stresses around the weld area. These dangers are minimal when the metals being joined are thick. But they may become problems when the base metals are thin sections. High temperatures are expensive as well since heat is energy, and energy costs money. The more heat you need to make the joint, the more the joint will cost to produce. Now consider the automated process.
What happens when you join not one assembly, but hundreds or thousands of assemblies. Welding, by its nature, presents problems in automation. We know that a resistance weld joint made at a single point is relatively easy to automate. But once the point becomes a line - a linear joint - the line has to be traced. It's possible to automate this tracing operation, moving the joint line, for example, past a heating station and feeding filler wire automatically from big spools. But this is a complex and exacting setup, warranted only when you have large production runs of identical parts.
Of course, welding techniques continually improve. You can weld on a production basis by electron beam, capacitor discharge, friction and other methods. But these sophisticated processes usually call for specialized and expensive equipment and complex, time consuming setups. They're seldom practical for shorter production runs, changes in assembly configuration or - in short - typical day-to-day metal joining requirements.
Oxyacetylene can be used to braze and to braze weld (bronze weld). These techniques are often confused, as they involve the use of the same equipment.
Bronze filler rod is an alloy composed of copper and tin. Brass filler rod is also an alloy, composed of copper and zinc. Either rod can be used.
The steel tube and steel plate have been heated to a dull red heat. The ‘bronze’ filler rod (coated with flux) is then introduced to the joint. It flows onto the two surfaces, forming a ‘fillet’ around the joint, joining the two surfaces.
Braze / bronze welding provides a strong joint and is ideal of steel frames, where a certain amount of flexibility is required.
The filler rod is broader than that used in brazing.
When brazing, flux is applied to the joint, to prevent oxidation of the surfaces. The steel tube and plate are heated to bright red heat and the brazing rod (filler rod) applied to the joint. The end of the filler rod melts and flows around the joint, through capillary attraction.
This gives a joint that looks like a ‘line’ of bronze.
Brazing is ideal for manufacture of bicycle frames, as it does not ‘fracture’ as easily as steel welded joints.
Brazing and braze welding (bronze welding), can be used to join similar and also dissimilar metals such as:
Mild Steel - Galvanized Steel
Stainless Steel - Copper
Mild Steel - Stainless Steel
Stainless Steel - Copper-Nickel
Mild Steel - Cast Iron
Basics of flame brazing and soldering
Soldering and brazing are joining processes whereby a filler metal or alloy is heated to melting temperature above its liquid temperature but below the melting point of the metals being joined.
Traditionally, brazing and soldering are distinguished from each other in the following manner:
Joining method
Soldering and brazing are joining processes whereby a filler metal or alloy is heated to melting temperature above its liquid temperature but below the melting point of the metals being joined.
Traditionally, brazing and soldering are distinguished from each other in the following manner:
- Flame soldering of aluminium: TL < 450 °C
- Flame brazing of aluminium: TL > 450 °C
- Joint clearances
- Cleanness
- Selection of flux and filler material
Joining method
Fluxes
Fluxes are chemical compounds applied to the joint surfaces before brazing.
Their use is essential in the soldering/brazing processes. Heating a metal surface accelerates the formation of oxides, the result of chemical combination between the hot metal and oxygen in the air. This oxide layer has to be removed before brazing, and the formation of a new oxide layer has to be prevented or they will inhibit the filler metal from wetting and bonding to the surfaces.
Fluxes are chemical compounds applied to the joint surfaces before brazing.
Their use is essential in the soldering/brazing processes. Heating a metal surface accelerates the formation of oxides, the result of chemical combination between the hot metal and oxygen in the air. This oxide layer has to be removed before brazing, and the formation of a new oxide layer has to be prevented or they will inhibit the filler metal from wetting and bonding to the surfaces.
Generally, fluxes play the following roles:
Non-corrosive potassium flux paste for Al-Si brazing
Non-corrosive flux paste (based on potassium fluoroaluminate salts KAlF4 K3AlF6) can be used with filler AA4047 in a controlled atmosphere furnace, torch, or induction brazing processes for joining a wide variety of aluminium alloys. Adding potassium to the conventional fluoride fluxes allows for better brazing of aluminium magnesium alloys such as 6061 and 6063. No post-braze cleaning operations are required. Potassium flux paste and its residues are non-hygroscopic and non-corrosive.
Non-corrosive caesium flux paste for Al-Zn soldering
Special flux paste with caesium aluminium salts (CsAlF4) and a proprietary binder are available to be used in atmosphere, furnace, torch or induction brazing processes for joining a wide variety of aluminium alloys using low temperature filler alloys of zinc/aluminium that performs in the temperature range from 382-to-500°C. The flux and its residues are non-hygroscopic and non-corrosive.
- Fluxes dissolve surface oxides
- Fluxes permit faying surfaces to wet
- Fluxes encourage filler metal flow
- Fluxed prevent re-oxidation during the brazing process
- Non-corrosive potassium flux paste for Al-Si brazing
- Non corrosive caesium flux paste for Al-Zn soldering.
Non-corrosive potassium flux paste for Al-Si brazing
Non-corrosive flux paste (based on potassium fluoroaluminate salts KAlF4 K3AlF6) can be used with filler AA4047 in a controlled atmosphere furnace, torch, or induction brazing processes for joining a wide variety of aluminium alloys. Adding potassium to the conventional fluoride fluxes allows for better brazing of aluminium magnesium alloys such as 6061 and 6063. No post-braze cleaning operations are required. Potassium flux paste and its residues are non-hygroscopic and non-corrosive.
Non-corrosive caesium flux paste for Al-Zn soldering
Special flux paste with caesium aluminium salts (CsAlF4) and a proprietary binder are available to be used in atmosphere, furnace, torch or induction brazing processes for joining a wide variety of aluminium alloys using low temperature filler alloys of zinc/aluminium that performs in the temperature range from 382-to-500°C. The flux and its residues are non-hygroscopic and non-corrosive.
google-site-verification: google231c230fc8f05942.html